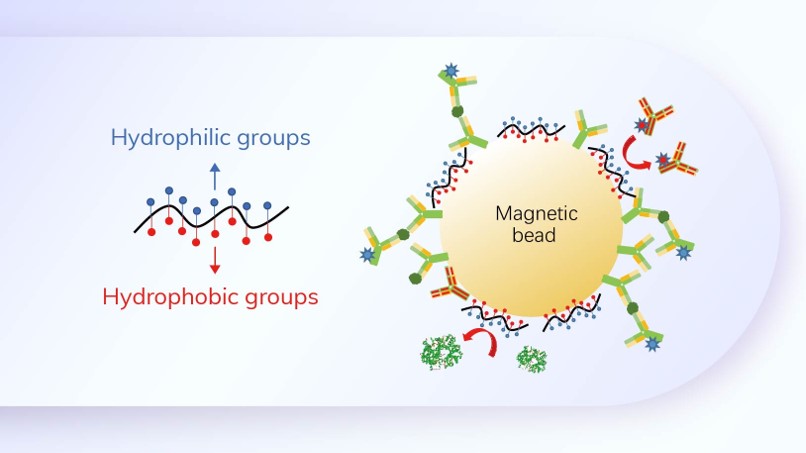
Innovative R&D
Innovative R&D
Fapon Biotech places great importance to
independent R&D, making continuous breakthroughs and iteration. We have
world-class R&D and application platforms, rich experiences in reagent
material labeling, matching and screening, reagent development debugging and
performance optimization, with an excellent technology development process
implement to meet customer needs precisely.