Related Suggestion
Fapon Biotech, a mainstream COVID-19 raw materials supplier to the global top-profile reagent manufacturers, introduced pioneering COVID-19 antibodies for antigen lateral flow testing to detect and differentiate COVID-19 and the most notable variant B.1.1.7 (also known as SARS-CoV-2 VUI 202012/01) in human samples.
Variant strain surveillance is vital for COVID-19 control. Variants such as B.1.1.7 that causes 40-70% higher transmissibility and false-negative results on some PCR tests had drawn concerns and investigation from scientists and healthcare authorities like WHO, FDA and others. According to WHO’s report on December 14, at least 50 countries had confirmed the identification of B.1.1.7 cases.
The market demand for COVID-19 variant testing was once determined and anticipated by Fapon Biotech R&D team upon revealing the UK variant genome sequence. The company took the R&D approach from a different angle, adopted the idea of reverse technology design, and rapidly introduced the technology with a unique principle that can be used widely to identify variant strains.
The launch of the product and technology took Fapon Biotech’s corporate mission, ‘Enable Earlier Disease Identification, More Convenient, Accurate and Affordable Diagnosis’ a step further in COVID-19 strain surveillance, adding great addition to current variant strain genome sequencing and PCR testing technologies. Fapon Biotech will continue keeping pace with changing trends in COVID-19 diagnosis and innovate to roll out products to help customers unlock market opportunities.
First in World, COVID-19 Antibodies for Antigen Lateral Flow Testing in Differentiating Variant B.1.1.7 and Wild-Type Virus
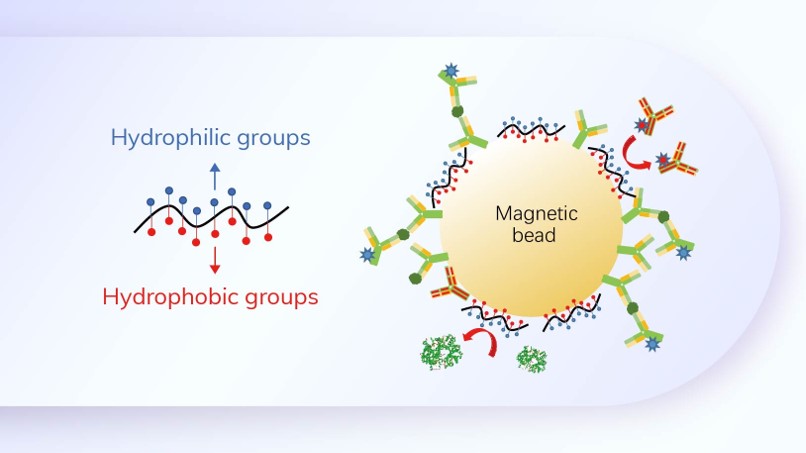
Principle
An innovative REVERSE technology design that uses two different antibodies for wild-type and B.1.1.7 variant strains detection
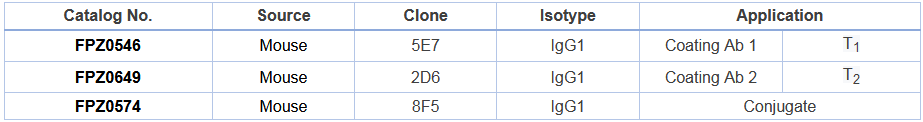
l T1 Monoclonal antibodies for all COVD-19 viral strain detection
l T2 Monoclonal antibodies for non-detection or a very weak detection of B.1.1.7 variant strain, but detectable to other COVD-19 viral strains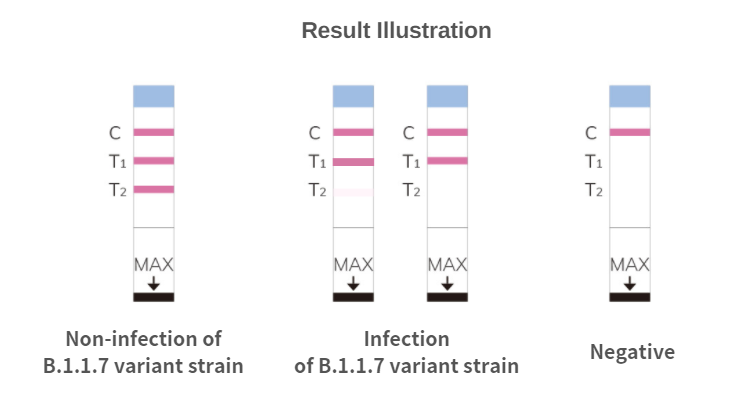
Product Information
Performance
Activity: antibody pairs can effectively detect wild-type recombinant antigens, B.1.1.7 recombinant antigens and other main N-protein mutated recombinant antigens
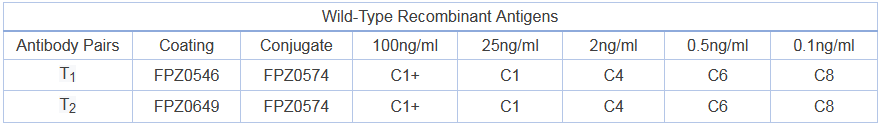
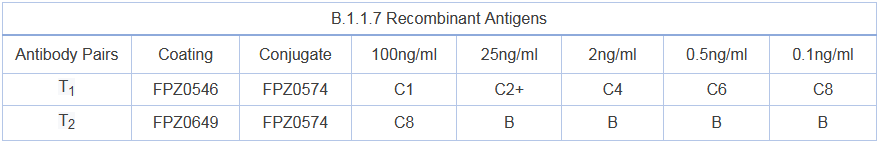
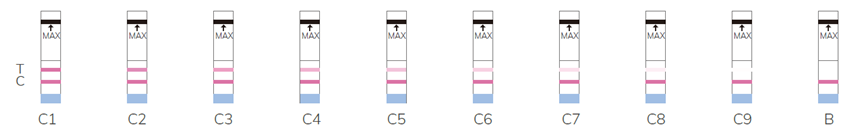
Note: the higher the number, the lower the activity, B refers to undetectable
Specificity: no false-positive results shown in the detection of 100 healthy human nasal swab samples and 100 healthy human throat swab samples
* Please note that performances presented above were based on the recombinant antigen evaluations, Fapon Biotech will continue to conduct the next-step clinical sample verification.